Problems related to pH and practical solutions
We all know that the optimal pH level for most crops is between 6 and 7.5. But what do other values mean?
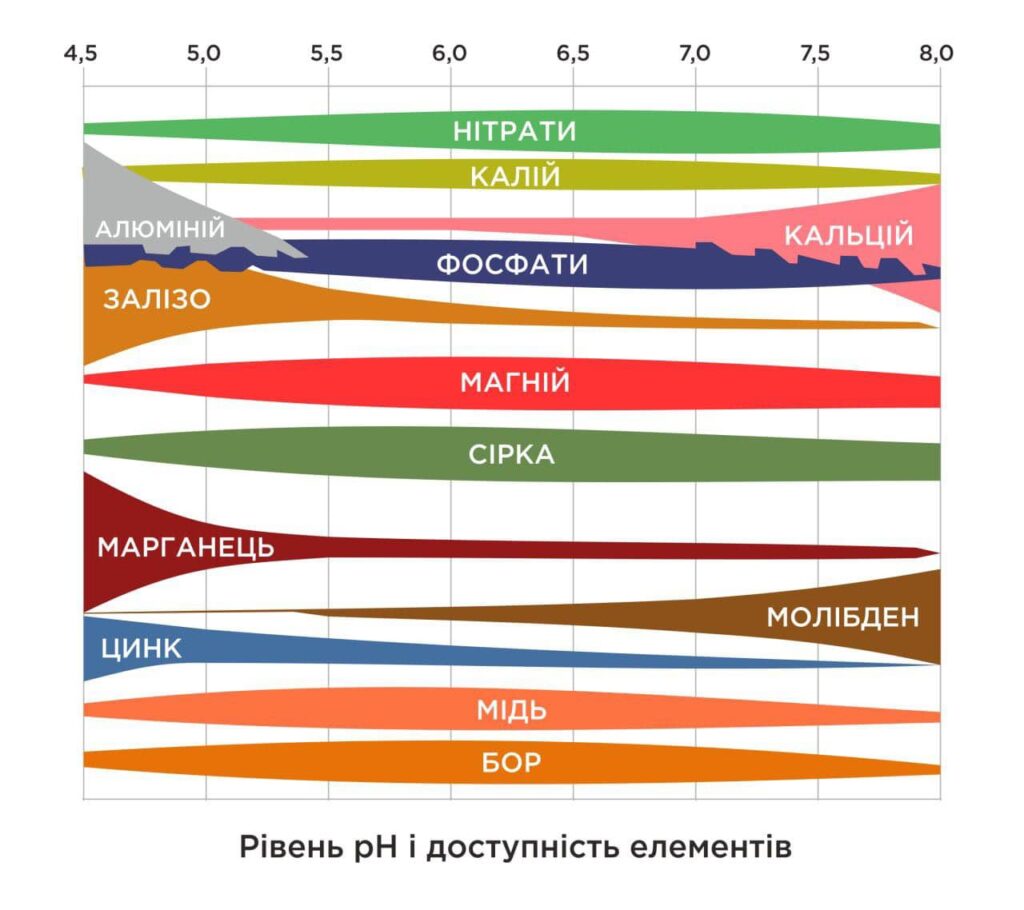
How pH affects yields:
Low pH < 7 (acidic soil):
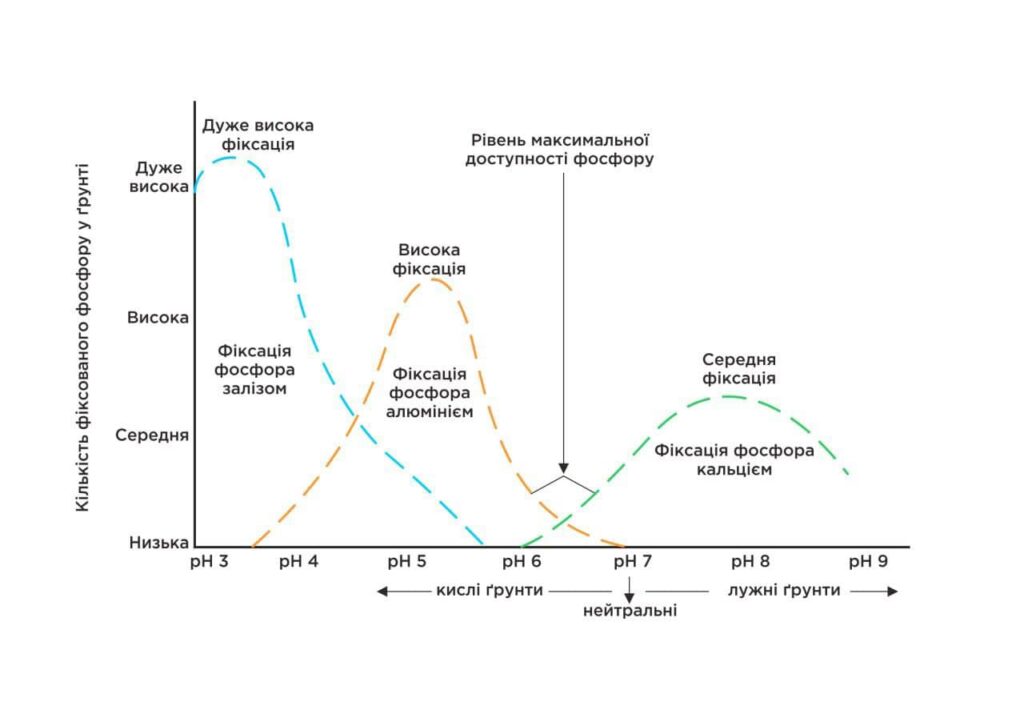
- Phosphorus is maximally available up to 5.8. pH < 5.4 – phosphorus binds to iron and aluminium and becomes less available
- Trace elements (iron and manganese) may be present in excess, potentially toxic to plants.
- Calcium and magnesium become deficient, which affects plant health.
High pH > 7 (alkaline soil):
- Iron, zinc, copper and boron become less available due to the formation of insoluble bases or basic salts, which can cause leaf chlorosis.
Ways to adjust the pH:
To increase the pH of acidic soils:
- Lime application: Neutralises acidity, improves calcium and magnesium availability.
- Application of dolomite flour: Enriches the soil with calcium and magnesium, increases the pH.
To lower the pH of alkaline soils:
- Use of ammonium sulphate: Provides nitrogen and lowers pH, improving micronutrient availability.
- Addition of elemental sulphur: Lowers pH, improves availability of iron, copper, and other trace elements.
- In addition, as the activity of soil microorganisms increases, a lot of carbon dioxide is released into the soil, which leads to a decrease in pH.
General recommendations:
- Apply phosphate fertiliser close to the root of the plants to ensure better absorption.
- Enrich with organic materials – this improves soil structure, reduces erosion and nutrient leaching.
Test the soil regularly to help detect changes in the pH level and correct them in time.

Write to us
and we will find an opportunity
for cooperation


